Exploring chaos on the nanometer scale
Chaotic behavior is typically known from large systems: for example, from weather, from asteroids in space that are simultaneously attracted by several large celestial bodies, or from swinging pendulums that are coupled together. On the atomic scale, however, one does normally not encounter chaos—other effects predominate.
混沌行为通常来自大型系统:例如,来自天气,来自同时被几个大天体吸引的太空中的小行星,或来自耦合在一起的摆动钟摆。然而,在原子尺度上,人们通常不会遇到混沌——其他效应占主导地位。
Now, for the first time, scientists at TU Wien have been able to detect clear indications of chaos on the nanometer scale—in chemical reactions on tiny rhodium crystals. The results have been published in the journal Nature Communications.
现在,维也纳理工大学的科学家们第一次能够在纳米尺度上探测到混沌的清晰迹象——在微小的铑晶体上发生化学反应。研究结果已发表在《自然通讯》杂志上。
From inactive to active—and back again
从不活跃到活跃,然后再回来
The chemical reaction studied is actually quite simple: with the help of a precious metal catalyst, oxygen reacts with hydrogen to form water, which is also the basic principle of a fuel cell. The reaction rate depends on external conditions (pressure, temperature). Under certain conditions, however, this reaction shows oscillating behavior, even though the external conditions are constant.
研究的化学反应其实很简单:在贵金属催化剂的帮助下,氧与氢反应生成水,这也是燃料电池的基本原理。反应速率取决于外部条件(压力、温度)。然而,在某些条件下,即使外部条件是恒定的,该反应也会表现出振荡行为。
“Similar to the way a pendulum swings from left to right and back again, the reaction rate oscillates between barely perceptible and high, and thus the catalytic system oscillates back and forth between inactive and active states,” explains Prof. Günther Rupprechter from the Institute of Materials Chemistry at TU Wien.
维也纳理工大学材料化学研究所的Günther Rupprechter教授解释说:“类似于钟摆从左到右再摆回来的方式,反应速率在几乎察觉不到和很高之间振荡,因此催化系统在无活性和活性状态之间来回振荡。”
A pendulum is a classic example of something predictable—if you disturb it a bit or set it in motion twice in slightly different ways, it behaves broadly the same. In this sense, it is the opposite of a chaotic system, where minimal differences in the initial conditions lead to strongly differing results in the long-term behavior. A prime example of this behavior are several pendulums connected by elastic bands.
钟摆是可预测事物的一个经典例子——如果你稍微打乱它,或者以稍微不同的方式让它运动两次,它的表现基本上是一样的。在这个意义上,它与混沌系统相反,在混沌系统中,初始条件的微小差异会导致长期行为的强烈不同结果。这种行为的一个主要例子是几个由橡皮筋连接的钟摆。
Setting exactly the same initial conditions twice is impossible
两次设置完全相同的初始条件是不可能的
“In principle, of course, laws of nature still determine exactly how pendulums behave,” says Prof. Yuri Suchorski (TU Wien). “If we could start such a coupled system of pendulums in exactly the same way twice, the pendulums would move exactly the same way both times.”
“当然,从原则上讲,自然法则仍然确切地决定了钟摆的行为,”Yuri Suchorski教授(维也纳理工大学)说。“如果我们能以完全相同的方式启动这样一个钟摆耦合系统两次,钟摆就会两次以完全相同的方式运动。”
But in practice, that’s impossible: you’ll never be able to perfectly recreate the same initial situation the second time as you did the first—and even a vanishingly small difference in the initial conditions will cause the system to behave completely different than the first time—this is the famous “butterfly effect”: small differences in the initial conditions lead to huge differences in the state at a later time.
但在实践中,这是不可能的:你永远不可能在第二次中完美地重现与第一次相同的初始状态——即使初始条件的微小差异也会导致系统的行为与第一次完全不同——这就是著名的“蝴蝶效应”:初始条件的微小差异会导致以后状态的巨大差异。
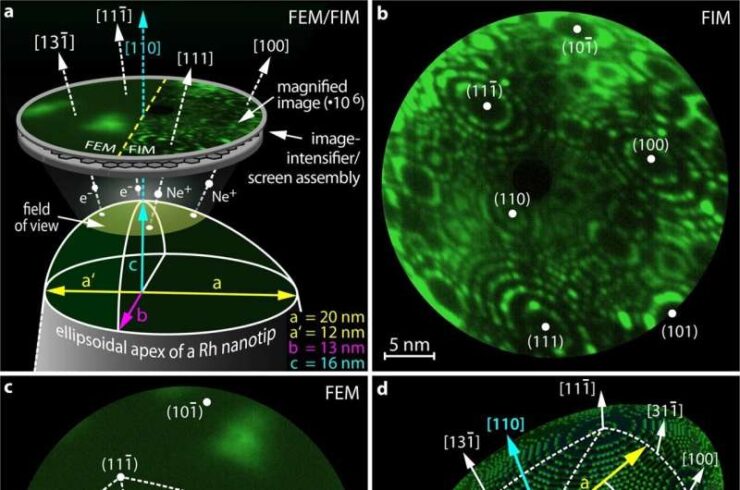
Something very similar has now been observed during chemical oscillations on a rhodium nanocrystal: “The crystal consists of many different surface nanofacets, like a polished diamond, but much smaller, on the order of nanometers,” explain Maximilian Raab and Johannes Zeininger, who performed the experiments. “On each of these facets, the chemical reaction oscillates, but the reactions on neighboring facets are coupled.”
现在在铑纳米晶体的化学振荡中观察到非常相似的现象:“晶体由许多不同的表面纳米层面组成,就像抛光的钻石一样,但要小得多,大约在纳米级,”进行实验的马克西米兰·拉布和约翰内斯·蔡宁格解释说。“在每一个层面上,化学反应都在振荡,但相邻层面上的反应是耦合的。”
Switching—from order to chaos
从有序到混乱
The coupling behavior can now be controlled in a remarkable way—by changing the amount of hydrogen. Initially, one facet dominates and sets the pace like a pacemaker. All other facets join in and oscillate to the same beat. If one increases the hydrogen concentration, the situation becomes more complicated. Different facets oscillate with different frequencies—but still their behavior is periodic and well predictable.
这种耦合行为现在可以通过改变氢的数量来控制。最初,一个方面占主导地位,并像起搏器一样设定节奏。所有其他方面都加入进来,并以相同的节奏振荡。如果增加氢的浓度,情况会变得更复杂。不同的面以不同的频率振荡,但它们的行为仍然是周期性的,并且很好地可预测。
However, if one then increases the hydrogen concentration further, this order suddenly breaks down. Chaos wins, the oscillations become unpredictable, small differences in the initial situation lead to completely different oscillation patterns—a clear sign of chaos.
然而,如果进一步增加氢的浓度,这个顺序就会突然被打破。混沌获胜,振荡变得不可预测,初始情况的微小差异导致完全不同的振荡模式——这是混沌的明显迹象。
“This is remarkable because you wouldn’t really expect chaotic behavior in nanometer-sized structures,” says Yuri Suchorski. “The smaller the system, the greater the contribution of stochastic noise. In fact, the noise, which is something completely different from chaos, should dominate the behavior of the system: it is even more interesting that it was possible to ‘extract’ indications of chaos“. A theoretical model was particularly useful, developed by Prof. Keita Tokuda.
“这是值得注意的,因为你不会真正期望在纳米尺寸的结构中出现混沌行为,”Yuri Suchorski说。“系统越小,随机噪声的贡献就越大。事实上,噪声与混沌完全不同,它应该主导系统的行为:更有趣的是,有可能‘提取’混沌的迹象。”德田凯塔(Keita Tokuda)教授开发的一个理论模型特别有用。